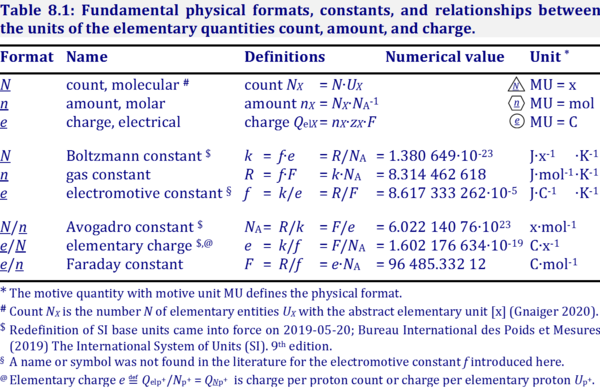
1 mol of charge = 1 Faraday = 96500 coulombs. Represent the cell and the standard emf of the cell having the following reaction. 2Cr(s) +3Ca(aq) = 2Cr + (aq) + 3Cd()

SOLVED: TABLE 13-1: Some Physical Constants and Units Used in Thermodynamics Boltzmann constant, k = 1.381 x 10^(-23) J/K Avogadro's number, N = 6.022 x 10^(23) mol^(-1) Faraday constant, F = 96,480
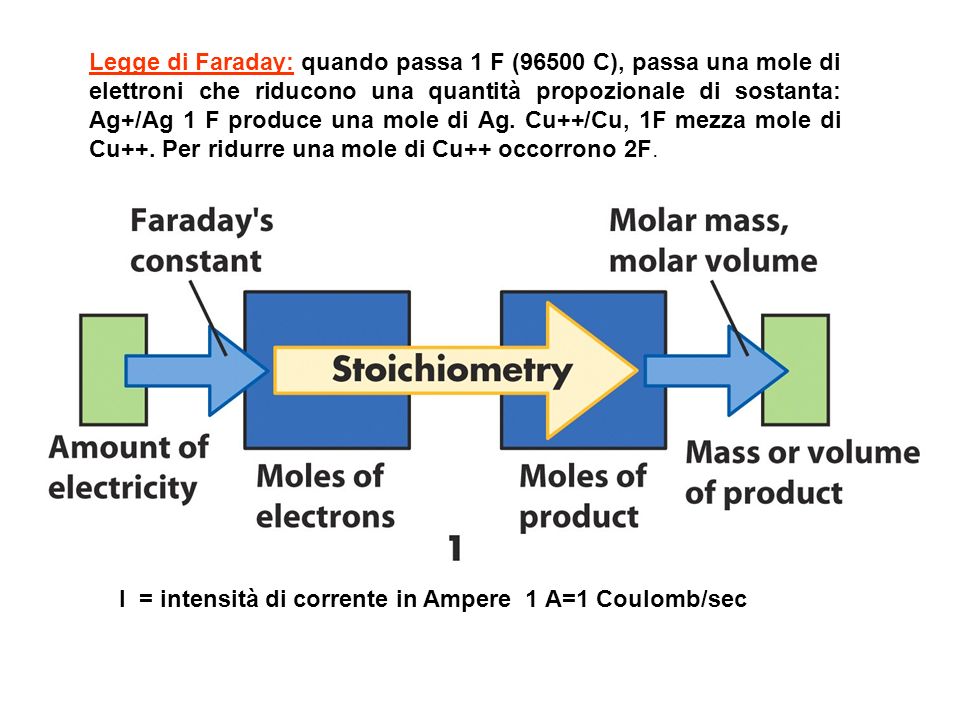
Legge di Faraday: quando passa 1 F (96500 C), passa una mole di elettroni che riducono una quantità propozionale di sostanta: Ag+/Ag 1 F produce una mole. - ppt scaricare
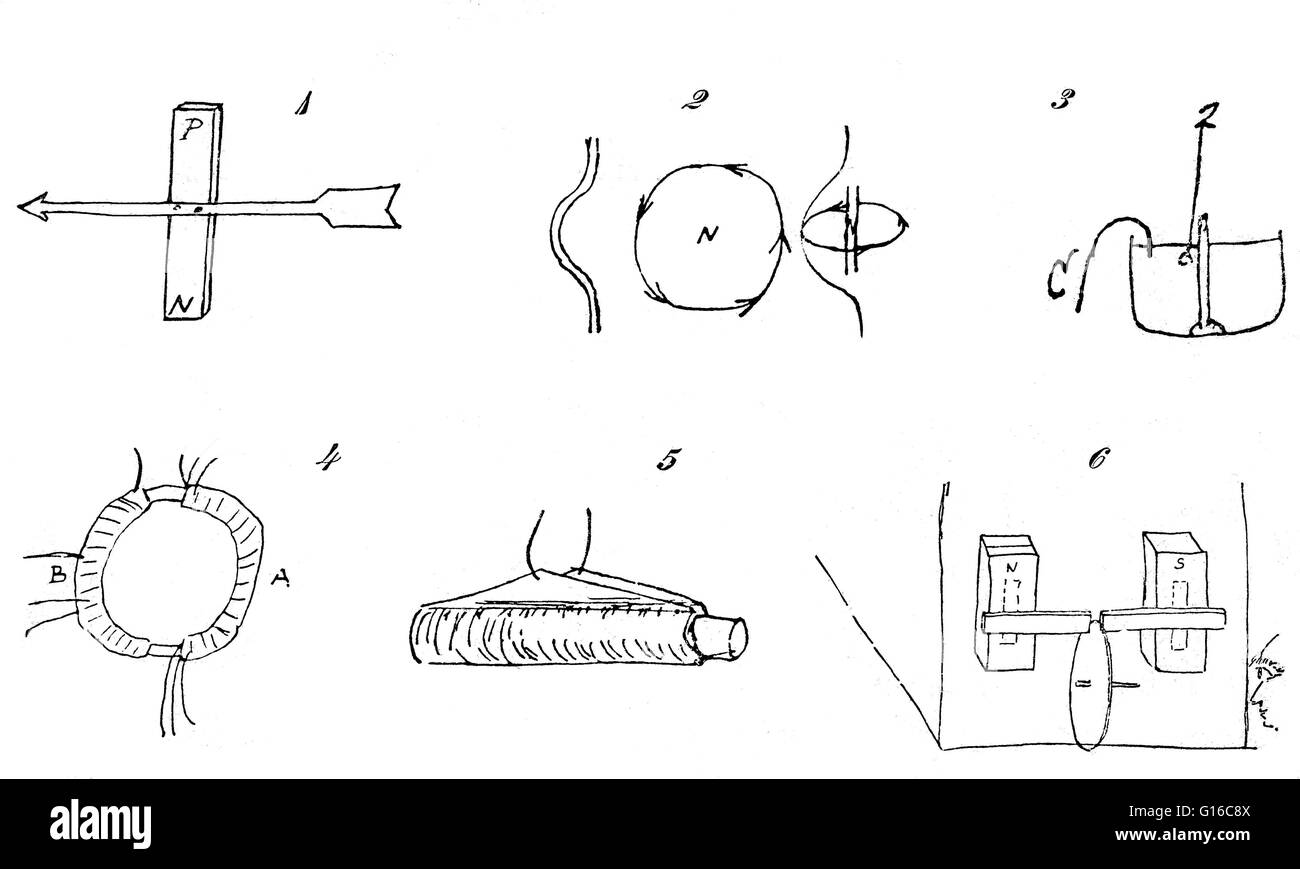
Schizzi da Faraday il diario della mostra la progressione dei suoi esperimenti elettromagnetici. 1) Egli ripete Oersted in seguito alla scoperta del 1820 che un ago magnetico eccepire nei pressi di un
1.Explain and give e.g. on 'how 1 Faraday will always deposits 1 gm equivalent mass of a substance.'

1 Suppose we had a 0.3A current producing 15mL of hydrogen gas in 6 min and 30 seconds. How much charge did we have per one mole of electrons? We need. - ppt download

The mass of the substance deposited by 1 faraday of electricity is equal to 11 grams. The value of electrochemical equivalent is: A. 11 B. 11 x 96500 - Correct Answers 11 96500 D. data insufficient

SOLVED:The faraday is a unit of charge frequently encountered in electrochemical applications and named for the British physicist and chemist Michael Faraday. It consists of 1 mole of elementary charges. Calculate the